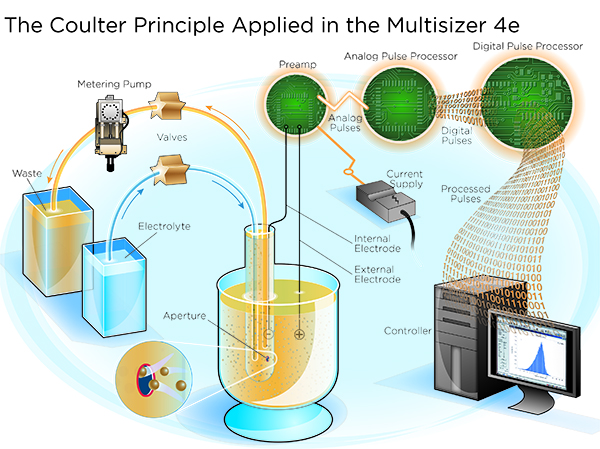
Technologies
We've been designing instruments since 1935 for labs, universities, hospitals and manufacturing facilities across the globe. Each of our instruments is designed with innovative technology at its core. Learn what the driving force is behind our instruments and reagents, and understand why scientists continue to trust Beckman products in their research.
Both life sciences and manufacturing in the 21st century encompass an amazing array of technologies that were considered science fiction only a few decades ago.
And those technologies will no doubt help inspire the creation of even more amazing technologies in the years ahead.
For an introduction to some of the technologies life scientists and manufacturers rely on today, you’re in the right place.