Methods for isolating extracellular vesicles
Whether from liquid biopsies or conditioned media, no other step is as critical to the success of your EV research as your choice of isolation technique. Differential ultracentrifugation has been called the “gold standard” method,(1) not only because it was the first method demonstrated to reliably isolate a pure fraction of EVs, but also because EVs produced by differential ultracentrifugation are largely intact and thought to contain a full complement of cargo molecules.
Other methods, like bead- or antibody-based protocols, require exosome-surface interactions that may damage the vesicle, diluting or depleting the vesicular cargoes.
As experts in ultracentrifugation, we have decades of experience—supported by products and protocols—to help enable your discovery.
EVs are secreted by most cells types and, therefore, may be isolated from cell culture as well as bodily fluids including plasma, urine, saliva, serum, and cerebrospinal fluid.(2)
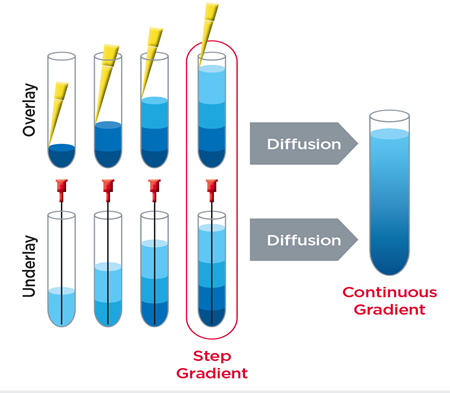
Protocols for the isolation of EVs have been tailored for each sample type by varying parameters such as the speed of centrifugation, use of filtration and density gradients, among other techniques.(3) One of the most widely practiced methods of isolating and purifying EVs from various sources is differential ultracentrifugation.(4) In this method, a series of spins facilitates the stepwise reduction of contaminants such as dead cells, cell debris, platelets, proteins, and nucleic acid complexes.(3)
Other techniques for EV isolation, often used in combination with differential centrifugation, include(5):
- Density gradient ultracentrifugation (DGUC)
- Precipitation
- Ultrafiltration
- Immunoaffinity isolation
- Size exclusion chromatography
- Microfluidics techniques
Each method has advantages and drawbacks, so attention should be given to selecting a method that best serves the downstream application.
In addition, some isolation and purification methods may produce high yields but render the vesicles non-viable for certain applications.(5) For example, size exclusion chromatography has been shown to produce highly pure samples with relatively low contamination, cost, and processing time. However, the final sample may be significantly diluted, throughput is low, and the resulting product is a broad population of EV sizes.(5)
The cons of contaminants
Non-EV content within a sample represents a significant challenge for the analysis of EV characteristics, as well as to their use in assays and other approaches.(3)
Some contaminants that can be difficult to remove from vesicle preparations include(3):
- Lipoprotein particles
- Microbes
- Microsomes
- Protein aggregates
- DNA and other products of necrosis
Depending on the intended downstream application of the purified EVs, further steps may be taken to remove contaminants from the purified sample. For example, it’s been demonstrated that EV preparations generated with an iodixanol density gradient achieve high purity, as evidenced by enrichment of the EV marker CD63 and absence of contaminating proteins, like extracellular Argonaute-2 complexes.(6)
A common method for estimating the amount of contaminating protein is by comparing the ratio of nano-vesicle counts (obtained through vesicle visualization technologies such as the NanoSight platform) to protein concentration (obtained with colorimetric protein assays such as the BCA assay).(6)
The presence of contaminating proteins alters this ratio and can therefore provide an estimate of the purity of the EV sample. It has been reported that samples with ratios greater than 3 x 1010 particles per µg protein are considered highly pure, while ratios of 2 × 109 to 2 × 1010 particles per µg protein are considered of low purity.(6)
When developing an isolation protocol, it is recommended that the sample be evaluated and optimized not only for the presence of EVs, but also for the absence of contaminating factors.
Comparison of EV Isolation Methods
| METHOD | BENEFITS | DISADVANTAGES |
| Differential Ultracentrifugation |
|
|
| Density Gradient Centrifugation |
|
|
| Size-exclusion Chromatography |
|
|
| Antibody-Coated Magnetic Beads |
|
|
| Microfluidic Devices |
|
|
| Precipitation |
|
|
Whichever isolation method you choose, there is an art to the science of EV isolation, in that more is not always better, and the best guide is good judgment and previous experience.
Using overly stringent size cutoffs or antigenicity requirements can be as detrimental as using overly broad size cutoffs or antigenicity requirements. The key to keep in mind is that sample purity and integrity are of the highest priority.
Maintaining a pure EV sample requires vigilance at all stages of the isolation procedure to prevent sample loss and to maintain the reliability of the data.
As is the case with any membrane-bound structure, from EV to animal cell, rough or improper handling may lead to lysis, and unintentional lysis of your EVs before you’re ready is the primary route to sample loss. To that end, be mindful of performing each step of your protocol at the recommended temperature and in the recommended buffer, while keeping your sample away from detergents and matrix metalloproteases.
Webinar: Nanoparticle Separations at Large Scale by C. Simone Plüisch and Alexander Wittemann
References
- Momen-Heravi F, Balaj L, Alian S, Mantel PY, Halleck AE, Trachtenberg AJ, et al. Current methods for the isolation of extracellular vesicles. Biol Chem. 2013;394(10):1253-62.
- Andaloussi SEL, Mager I, Breakefield XO, Wood MJ. Extracellular vesicles: biology and emerging therapeutic opportunities. Nat Rev Drug Discov. 2013;12(5):347-57.
- Witwer KW, Buzas EI, Bemis LT, Bora A, Lasser C, Lotvall J, et al. Standardization of sample collection, isolation and analysis methods in extracellular vesicle research. J Extracell Vesicles. 2013;2.
- Thery C, Amigorena S, Raposo G, Clayton A. Isolation and characterization of exosomes from cell culture supernatants and biological fluids. Curr Protoc Cell Biol. 2006; Chapter 3: Unit 3 22.
- Van Deun J, Mestdagh P, Sormunen R, Cocquyt V, Vermaelen K, Vandesompele J, et al. The impact of disparate isolation methods for extracellular vesicles on downstream RNA profiling. J Extracell Vesicles. 2014;3.
- Webber J, Clayton A. How pure are your vesicles? J Extracell Vesicles. 2013;2.

